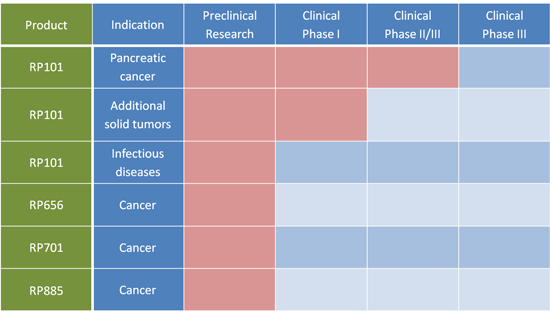
RP101 is the Company's first small molecule drug candidate. RP101 is currently being evaluated in combination with standard chemotherapy to treat patients with pancreatic cancer. To date patients receiving RP101 combination therapy have shown significant improvement in median survival when compared to well matched historical controls.
Building on the successful discovery and clinical development of RP101 to date, the Company has synthesized and screened more than two hundred follow on compounds for anti-chemoresistance efficacy. The most promising compounds have demonstrated a five- to tenfold greater potency to inhibit chemoresistance when compared to RP101. All follow up compounds selected for further developments are proprietary and all corresponding patents have been filed.